IP basics in Life sciences
Did the title “Generic Drugs Can Be Denied Approval?!” surprise you?
This topic is connected to the so-called “patent linkage” system that I briefly mentioned in
Vol. 5,
“Do They Really Creep In? – Development Schedules of Generics and Biosimilars,” in the section on “Application, Review, and Approval.”
When a generic drug application is submitted, the Ministry of Health, Labour and Welfare (MHLW) examines not only pharmaceutical aspects—such as stability and equivalence to the originator drug—but also the relationship between the generic and the originator’s patents. If the Ministry determines that approving the generic “as is” would pose patent-related concerns, the generic will not be approved, even if it has met all pharmaceutical requirements.
This system lies at the heart of this column’s title. Readers may be surprised to learn that the MHLW, a pharmaceutical regulator, also considers patent issues. In this article, I would like to explore how patent linkage works in Japan.
Wait What? Generic Drugs Can Be Denied Approval?!
― What Is Patent Linkage? ―
What is Patent Linkage?
Patent linkage, broadly speaking, refers to the process of verifying the relationship between a generic drug and the patents covering the corresponding originator drug during regulatory review.
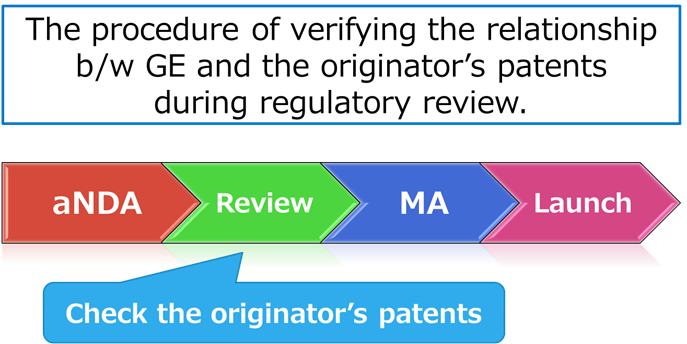
Did you know that this kind of “check” takes place during the review of generic drugs?
Those of you familiar with the Pharmaceuticals and Medical Devices Act (PMD Act) or the Patent Act might be thinking, “I’ve never heard of such a provision,” and you would be absolutely right. Patent linkage is not stipulated in the PMD Act, which is administered by the pharmaceutical authority, nor in the Patent Act, which is administered by the patent authority. Surprising, isn’t it?
Who Started Patent Linkage?
Patent linkage originated in the United States in 1984. Because it involves checking for potential patent infringement within the drug regulatory process, the system sits at the intersection of pharmaceutical regulation and patent administration. Since these authorities operate independently in any country, it is not easy for them to collaborate in creating legislation.
Against this backdrop, the United States sought to promote the overall development of its pharmaceutical industry by balancing the interests of originator and generic manufacturers. Senator Orrin Hatch and Representative Henry Waxman drafted legislation with this goal in mind. In 1984, patent linkage was enacted as the Hatch–Waxman Act, named after the two lawmakers.
Since then, several countries have introduced U.S.-style patent linkage after being requested to do so through free trade agreements (FTAs) with the United States.
Why Did Japan Introduce Patent Linkage?
Japan did not simply copy the U.S. approach. Its own system has been in place since the mid-1990s through notifications issued by the MHLW with the aim of ensuring a stable supply of pharmaceuticals. The current framework is defined in the following two notifications issued in October and November 2025:
- Notification dated October 8, 2025:
https://www.mhlw.go.jp/content/001575895.pdf - Notification dated November 14, 2025:
https://www.mhlw.go.jp/content/001594987.pdf
What Does Japan’s Patent Linkage System Look Like?
In Japan, patent linkage operates in two stages: (1) during the MHLW’s review of generic drug applications, and (2) in the period between approval of the generic and its listing on the National Health Insurance (NHI) drug price list.
First Stage: In the first stage, the MHLW examines the relationship between the generic drug and the patent information listed in the Pharmaceutical Patent Information Sheet (“Info Sheet”) submitted by the originator company when it applied for approval of the new drug. The Info Sheet covers both substance patents and use patents.
Pharmaceutical Patent Information Sheet
Originator companies are required to submit patent information related to the originator drug—substance patents and use patents are mandatory—when filing their new drug application. Additional patent information may be added until the end of the re-examination period. Furthermore, if a patent is granted after the re-examination period ends, the company must promptly add it to the Info Sheet.
Information on which patents were examined and how the MHLW evaluated them is not disclosed. If the Ministry determines that approving the generic would not pose a risk of infringing the originator’s patents, the generic is approved and becomes publicly known. However, if the Ministry concludes that approval would risk infringing the originator’s patents, the generic is simply not approved. In such cases, no formal “non-approval” decision is issued, and even the fact that the generic was filed remains hidden.
As a result, generic manufacturers often find themselves at a loss—unable to understand why their product was not approved or what they should do to obtain approval. None of this is publicly disclosed, and even the existence of generic companies struggling under these circumstances remains largely invisible. In recent years, however, some generic manufacturers have pursued litigation through various legal strategies. These cases have brought to light the fact that generic drugs are being denied approval because of patent linkage—a development that has attracted considerable attention.
Second Stage: The second stage is based on an MHLW notification that requests originator and generic companies to coordinate with each other and submit an NHI price listing application only for generics that are unlikely to infringe patents. This practice is commonly referred to as “pre-listing coordination.” Because the MHLW’s notification merely calls for companies to “conduct coordination,” even generics that complete this process and are successfully listed in the NHI price list may still become the subject of patent litigation.
How did you find this overview of patent linkage—a system that sits at the intersection of pharmaceutical regulation and patent law?
If you found yourself thinking, “Wouldn’t listing more substance and use patents in the Info Sheet make it harder for generics to enter the market?”—you have sharp instincts. Originator companies are well aware of this strategic angle.
With that in mind, next time I will take a closer look at how originator companies’ patent strategies have evolved. Stay tuned!
Author Profile

Yasuko Tanaka
President & Patent Attorney, S-Cube International Patent Office / CEO, S-Cube Corporation
Outside Director, Strategic Capital Inc.; Part-time Lecturer, Tokyo University of Agriculture and Technology Graduate School; Expert Commissioner in Intellectual Property Litigation
Previously served in the intellectual property departments of Teijin, Pfizer, and 3M Japan, with extensive experience in domestic and international IP practice, IP strategy development, contract negotiation, IP education, and project management. Founded S-Cube Corporation in April 2013 to strengthen global competitiveness of Japanese companies through IP strategy, and subsequently established S-Cube International Patent Office in August 2013 to provide seamless services including patent prosecution. Holds a B.Sc. in Biochemistry from Chiba University (1990).