“How a Single Mutation Can Change Fate — The Story of Hemoglobin” (Part 2)
A finely engineered giant: the structure of hemoglobin
Blood is red because red blood cells are densely packed with a protein called hemoglobin. It is the molecule that carries oxygen throughout the body—truly essential for sustaining life.
Let’s take a look at its structure (Figure 1).
Hemoglobin is a large complex made up of four subunits: two of one type of protein chain (the α chain) and two of another (the β chain), each with a very similar amino-acid sequence. Its molecular weight is about 66,000.
Each chain contains a heme iron, and oxygen binds to that site. By working together as a four-part assembly, hemoglobin can capture and release oxygen efficiently.
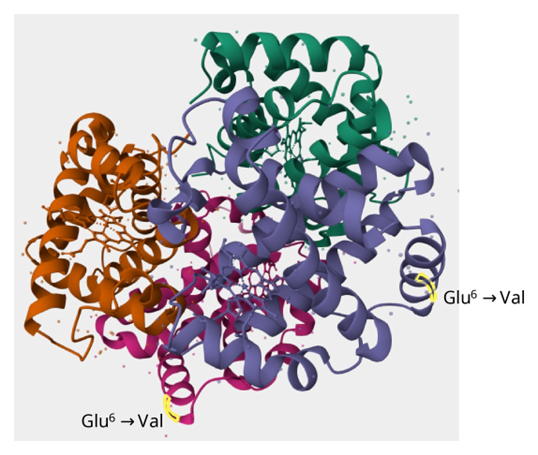
Ribbon model, PDB ID: 1A00. Hemoglobin consists of four subunits: two α chains and two β chains (blue and reddish-purple). One heme iron is coordinated in each subunit. The sixth amino acid of the β chain, shown in yellow, is Glu in healthy individuals, but is mutated to Val in carriers of sickle cell trait.
A difference in just one letter of the “code”…
However, a tiny “typo” can sometimes occur in the blueprint (gene) for this exquisitely designed hemoglobin.
When researchers examined hemoglobin from people with sickle cell anemia, they found that in the amino-acid sequence that forms the β chain, just one position—the 6th amino acid—had changed from glutamic acid (Glu) to valine (Val).
Out of hundreds of amino acids linked together, only one has changed.
Yet this small change dramatically alters the protein’s properties.
Red blood cells that change shape
When the 6th amino acid is replaced by valine, hemoglobin molecules become more likely to stick together when oxygen is released, forming long, fibrous structures.
As a result, red blood cells—which are normally soft and disc-shaped—become distorted as if propped from the inside by rigid rods, turning into a hard, sickle-like shape (Figure 2).
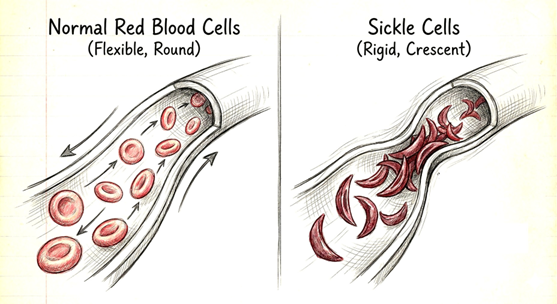
(Generated and modified using Gemini. An illustration of red blood cells deforming into a sickle shape due to mutant hemoglobin.)
These “sickled” red blood cells are fragile and break easily, leading to severe anemia. This is how a mutation at the molecular level can manifest as a visible disease.
Why this “mutation” was not eliminated
A gene that causes severe anemia would normally be expected to disappear through natural selection. Surprisingly, however, the distribution of this mutant gene closely overlaps with regions where malaria is prevalent (such as parts of Africa and South Asia).
Malaria is a life-threatening infectious disease in which parasites transmitted by mosquitoes invade red blood cells and cause high fever.
Because sickle-shaped red blood cells are fragile, they tend to rupture—often destroying the malaria parasites inside them as well. In other words, people with sickled red blood cells have strong resistance (tolerance) to malaria.
An “adaptive strategy” favored by evolution
Since genes are inherited one from each parent, we carry two copies of the same gene set.
In these regions, when one gene is normal and the other is the mutant type, individuals can gain both “blood that functions well enough for everyday life” and “resistance to malaria.”
Abnormal hemoglobin can certainly cause disease, but it can also be seen as a kind of “advantageous mutation” that humans acquired to survive in harsh environments.
There is no waste in each amino acid in a sequence—its presence reflects the outcome of natural selection over long periods of time.
Next time, under the theme “A world beyond genes — sophisticated structural changes and functional switching,” we will explore the drama that unfolds after an amino-acid sequence has been determined.
Proteins are not just simple strings. Depending on their environment and modifications, they can change shape with remarkable precision and even acquire entirely new functions.
Let’s explore the dynamic structural changes of proteins—and the mysteries of function that come with them—far beyond what the genetic code alone can ever fully explain.











